|
New England Biolabs
lambda monocut ladder 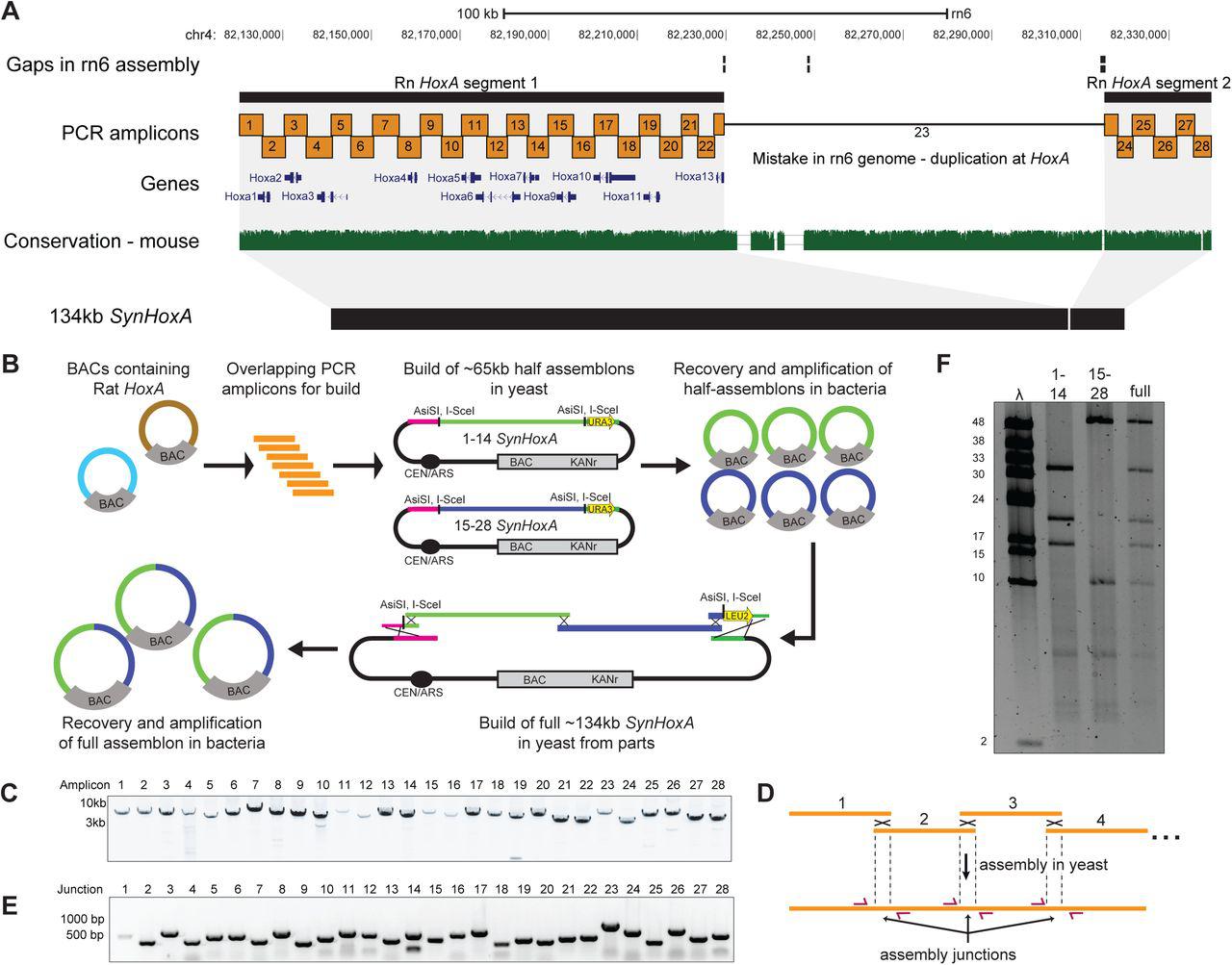 Lambda Monocut Ladder, supplied by New England Biolabs, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/lambda monocut ladder/product/New England Biolabs Average 93 stars, based on 1 article reviews
lambda monocut ladder - by Bioz Stars,
2026-06
93/100 stars
|
Buy from Supplier |
|
New England Biolabs
ladder lambda dna monocut mix  Ladder Lambda Dna Monocut Mix, supplied by New England Biolabs, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/ladder lambda dna monocut mix/product/New England Biolabs Average 93 stars, based on 1 article reviews
ladder lambda dna monocut mix - by Bioz Stars,
2026-06
93/100 stars
|
Buy from Supplier |
|
New England Biolabs
monocot λ mix  Monocot λ Mix, supplied by New England Biolabs, used in various techniques. Bioz Stars score: 96/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/monocot λ mix/product/New England Biolabs Average 96 stars, based on 1 article reviews
monocot λ mix - by Bioz Stars,
2026-06
96/100 stars
|
Buy from Supplier |
|
New England Biolabs
lambda dna  Lambda Dna, supplied by New England Biolabs, used in various techniques. Bioz Stars score: 96/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/lambda dna/product/New England Biolabs Average 96 stars, based on 1 article reviews
lambda dna - by Bioz Stars,
2026-06
96/100 stars
|
Buy from Supplier |
|
New England Biolabs
1 kb plus dna ladder  1 Kb Plus Dna Ladder, supplied by New England Biolabs, used in various techniques. Bioz Stars score: 97/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/1 kb plus dna ladder/product/New England Biolabs Average 97 stars, based on 1 article reviews
1 kb plus dna ladder - by Bioz Stars,
2026-06
97/100 stars
|
Buy from Supplier |
|
New England Biolabs
1 kb dna ladder  1 Kb Dna Ladder, supplied by New England Biolabs, used in various techniques. Bioz Stars score: 96/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/1 kb dna ladder/product/New England Biolabs Average 96 stars, based on 1 article reviews
1 kb dna ladder - by Bioz Stars,
2026-06
96/100 stars
|
Buy from Supplier |
|
New England Biolabs
hindiii  Hindiii, supplied by New England Biolabs, used in various techniques. Bioz Stars score: 99/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/hindiii/product/New England Biolabs Average 99 stars, based on 1 article reviews
hindiii - by Bioz Stars,
2026-06
99/100 stars
|
Buy from Supplier |
|
New England Biolabs
generuler dna ladder  Generuler Dna Ladder, supplied by New England Biolabs, used in various techniques. Bioz Stars score: 96/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/generuler dna ladder/product/New England Biolabs Average 96 stars, based on 1 article reviews
generuler dna ladder - by Bioz Stars,
2026-06
96/100 stars
|
Buy from Supplier |
|
Epicentre Biotechnologies
copy control library production kit  Copy Control Library Production Kit, supplied by Epicentre Biotechnologies, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/copy control library production kit/product/Epicentre Biotechnologies Average 90 stars, based on 1 article reviews
copy control library production kit - by Bioz Stars,
2026-06
90/100 stars
|
Buy from Supplier |
Image Search Results

Journal: bioRxiv
Article Title: Synthetic genomic reconstitution reveals principles of mammalian Hox cluster regulation
doi: 10.1101/2021.07.07.451065
Figure Lengend Snippet: (A) Layout of rat HoxA locus in the rn6 genome assembly. The rn6 genome includes an erroneous duplication at the HoxA locus between gaps in the assembly. The SynHoxA assemblon sequence is based on bringing together the two ‘separate’ RnHoxA segments. The sequence was segmented into 28 ∼5kb PCR amplicons with terminal homology of ∼200bp to adjacent amplicons. Conservation to the mouse genome is depicted using the multiz track from the UCSC genome browser. (B) Schematic depicting the assembly workflow for the 134kb SynHoxA assemblon. BACs containing Rat HoxA were used as PCR template to generate 28 segments tiling the entire HoxA locus. These segments were co-transformed into yeast with appropriate linkers and assembly vector to build two ∼65kb half assemblons into centromeric yeast-bacteria shuttle vectors. These half assemblons are recovered to bacteria and amplified. Full 134kb assemblon was built from half assemblons after releasing them from the vector using terminal restriction enzymes ( AsiSI ) and transforming into yeast. Full assemblon was then recovered from yeast into bacteria for amplification and verification. (C) Agarose gel of the 28 PCR amplicons that tile the 134kb SynHoxA assemblon. (D) Strategy to PCR-screen yeast colonies derived from assembly experiments. Primers (red arrows) span assembly junctions and test presence/absence of amplicons in many yeast colonies. Reproduced from ref with permission from authors. (E) Agarose gel showing one yeast colony carrying the full 134kb SynHoxA assemblon verified manually for the presence of all assembly junctions, using the strategy outlined in panel D. (F) Half and Full 134kb SynHoxA assemblon BACs purified from E.coli were digested with PvuI and separated using field inversion gel electrophoresis (FIGE). Lambda monocut ladder sizes are indicated in kb. Band sizes correspond to expected fragments.
Article Snippet: 500 ng of
Techniques: Sequencing, Transformation Assay, Plasmid Preparation, Amplification, Agarose Gel Electrophoresis, Derivative Assay, Purification, Nucleic Acid Electrophoresis

Journal: bioRxiv
Article Title: Synthetic genomic reconstitution reveals principles of mammalian Hox cluster regulation
doi: 10.1101/2021.07.07.451065
Figure Lengend Snippet: (A) Layout of rat HoxA locus from the rn6 genome assembly depicting genes, Rn HoxA cluster segments in black and previously identified distal enhancers in purple. The Enhancers+SynHoxA assemblon sequence is made by stringing all the enhancers directly upstream of the SynHoxA assemblon sequence. Conservation to mouse genome is depicted using multiz track from the UCSC genome browser. (B) PCR amplicons tiling enhancer sequences were generated from Rat HoxA BACs and co-transformed into a yeast strain containing the 134kb SynHoxA assemblon with a gRNA vector targeting the left terminus of the 134kb assemblon. The enhancer PCR amplicons were used to repair this break, resulting in the construction of the 170kb Enhancers+SynHoxA assemblon. Assemblon was recovered into bacteria for amplification and verification. (C) Agarose gel of the 8 PCR amplicons containing enhancer sequences. (D) Agarose gel showing one yeast colony tested for the presence of novel enhancer assembly junctions and with primers spanning 134kb SynHoxA . (E) 134kb and 170kb assemblon BACs purified from E.coli were digested with PvuI and separated using FIGE. Lambda monocut ladder sizes are indicated in kb. Band sizes correspond to expected fragments.
Article Snippet: 500 ng of
Techniques: Sequencing, Generated, Transformation Assay, Plasmid Preparation, Amplification, Agarose Gel Electrophoresis, Purification
Journal: bioRxiv
Article Title: Synthetic genomic reconstitution reveals principles of mammalian Hox cluster regulation
doi: 10.1101/2021.07.07.451065
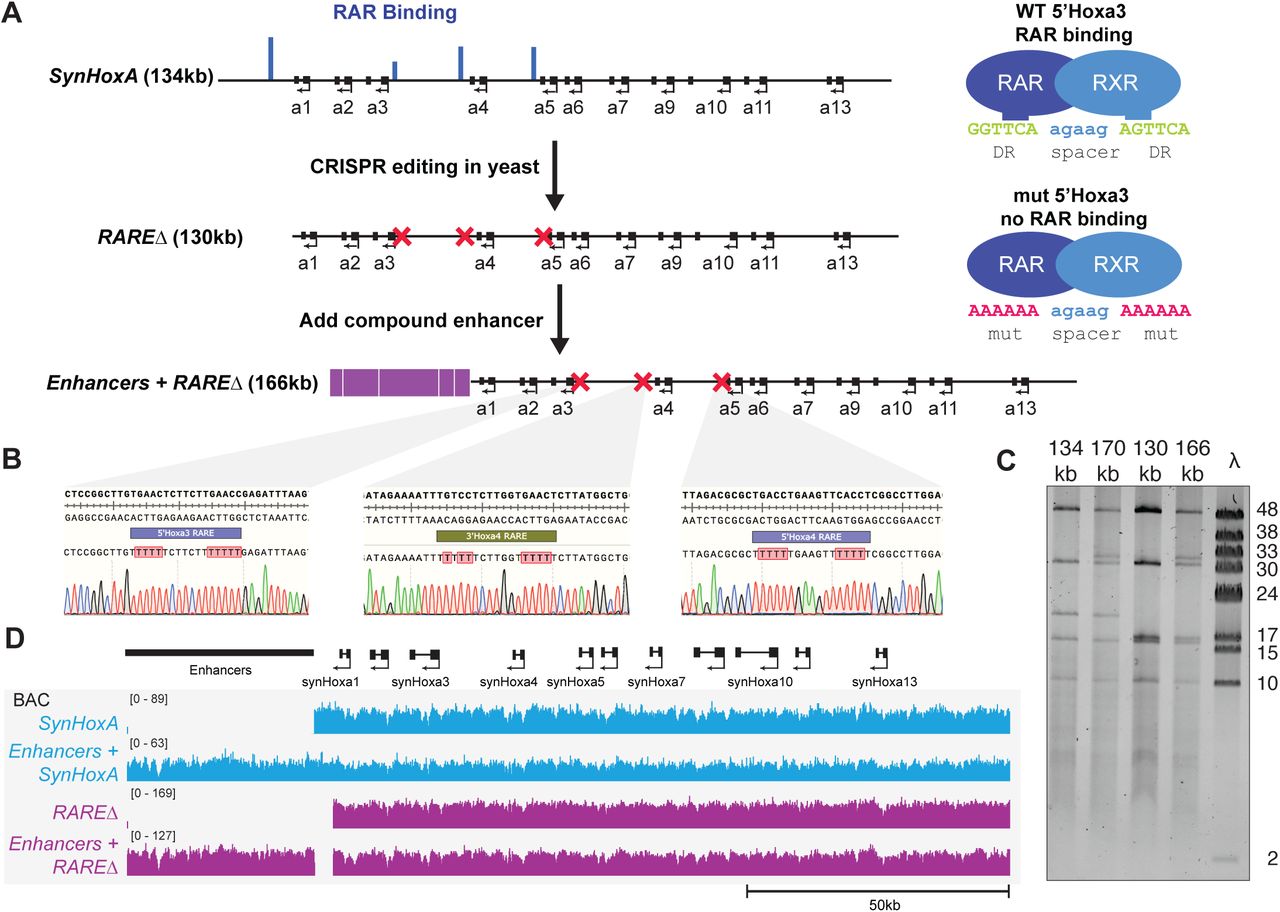
Figure Lengend Snippet: (A) Schematic of assembly strategy for 130kb RAREΔ SynHoxA and 166kb Enhancers + RAREΔ SynHoxA . Nature of the RARE mutations is shown on the right. RAR binding data comes from previously published reports. (see Methods) (B) Sanger sequencing traces confirmed precise CRISPR editing of RAREs in yeast. (C) SynHoxA assemblon BACs purified from E.coli were digested with PvuI and separated using FIGE. Lambda monocut ladder sizes are indicated in kb. Bands correspond to expected fragment lengths. (D) Sequencing data of assemblon BACs purified from E. coli aligned to a custom mm10 reference genome. Positions of the enhancers and protein coding genes are shown in black.
Article Snippet: 500 ng of
Techniques: Binding Assay, Sequencing, CRISPR, Purification